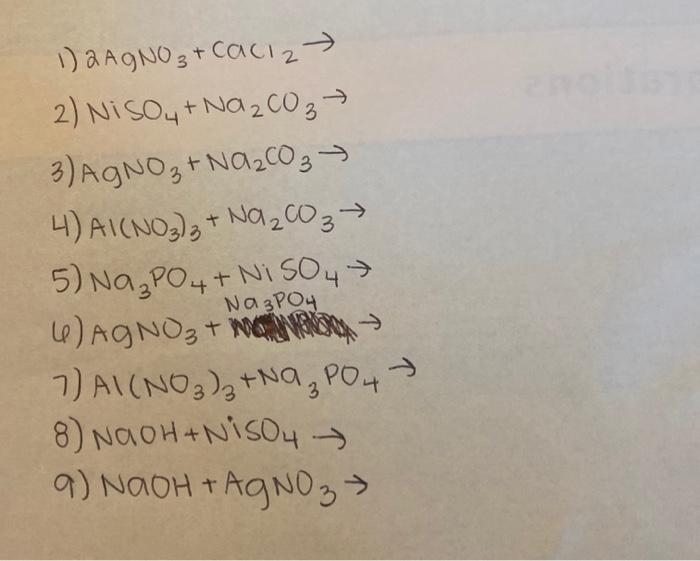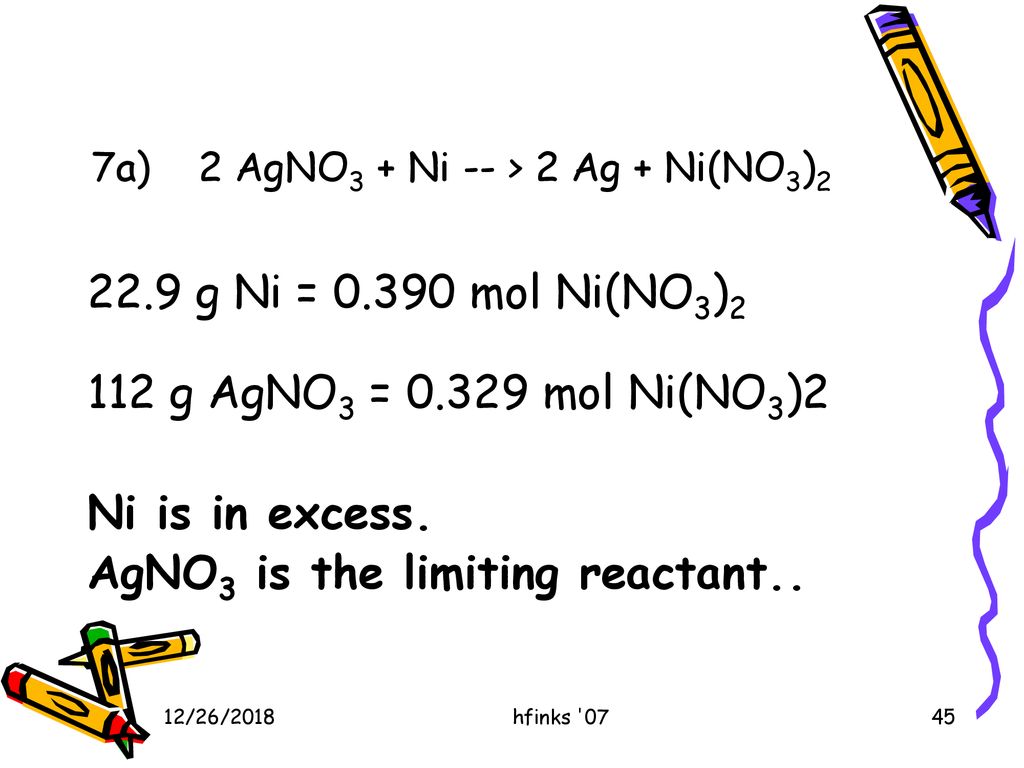A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal - Chemistry - Electrochemistry - 9165083 | Meritnation.com
69 Hydrocarbon (A), C6H10 on treatment with H2/Ni, H2/Lindlar catalyst and Na/liquid NH3 forms three different reduction products (B), (C) and (D) respectively. (A) does not form any salt with ammoniacal AgNO3

Scheme 1. Schematic illustration of the formation of Ag-Ni core-shell... | Download Scientific Diagram
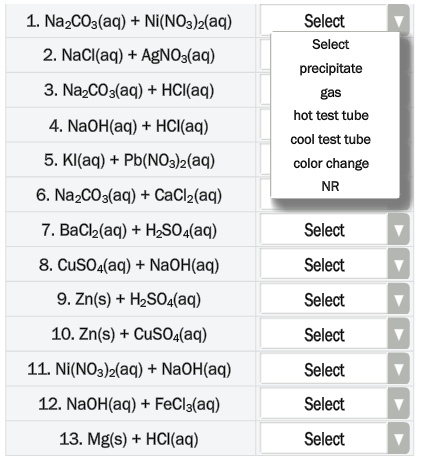
SOLVED: 1. Na2CO3(aq) + Ni(NO3)2(aq) 2. NaCl(aq) + AgNO3(aq) 3. Na2CO3(aq) + HCl(aq) â†' NaOH(aq) + CO2(g) + H2O(l) 4. KI(aq) + Pb(NO3)2(aq) â†' PbI2(s) + KNO3(aq) 5. Na2CO3(aq) + CaCl2(aq) â†'
![EXERCISE 11. Of the complex [Ni(NH3),Br]CI, the ionization isomer will give colour with AgNO3 (1) White (2) Red (3) Yellow (4) Blue 12. The compound PtCl2NH, does not react with AgNO. This EXERCISE 11. Of the complex [Ni(NH3),Br]CI, the ionization isomer will give colour with AgNO3 (1) White (2) Red (3) Yellow (4) Blue 12. The compound PtCl2NH, does not react with AgNO. This](https://toppr-doubts-media.s3.amazonaws.com/images/7410843/66afa896-6332-4a2e-a0c9-acc6154c6ca9.jpg)
EXERCISE 11. Of the complex [Ni(NH3),Br]CI, the ionization isomer will give colour with AgNO3 (1) White (2) Red (3) Yellow (4) Blue 12. The compound PtCl2NH, does not react with AgNO. This